© Aria Analytics, 2015

Advanced Liquid Analysis for Industry and Healthcare






A World of Information in a Drop
A 5iTech Company
Applicable in:
•
Milk
•
Wine
•
Beer •
Juices
•
Juices
 •
Juices
•
Juices
Healthcare •
IV Solutions
•
Dosage and Mix Control
•
IV Solutions
•
Dosage and Mix Control •
Counterfeit Medications
•
Counterfeit Medications •
Contaminated Pharmaceuticsals
•
Contaminated Pharmaceuticsals •
Medical Screening
•
Medical Screening
 •
IV Solutions
•
Dosage and Mix Control
•
IV Solutions
•
Dosage and Mix Control •
Counterfeit Medications
•
Counterfeit Medications •
Contaminated Pharmaceuticsals
•
Contaminated Pharmaceuticsals •
Medical Screening
•
Medical Screening
Better Quality ... Lower Cost ... Improved Safety
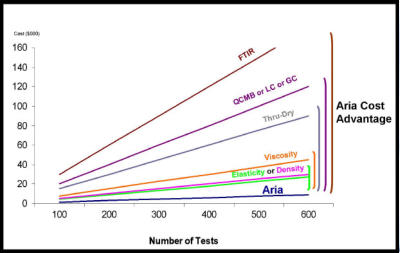
Almost any complex liquid sample can be characterized by
Aria Analytics’ novel acoustic technology. The same sensor is
used to test chemicals, coatings, drugs, and many other
products. Using a single sample and instrument, we capture
the sample’s unique “fingerprint” as the liquid undergoes its
predictable natural drying process. The results contain
important data related to its composition, physical and
chemical changes. Currently, similar data is available only by
running multiple tests on multiple analytical instruments
followed by skilled interpretation. The Aria holistic approach to
analysis lowers both capital and operating costs per test.
Quality Control
Inks
Due to its ability to detect even minute differences from one sample to another, the Aria technology is ideally suited to Quality Control and Quality Assurance.
Currently, any number of single analytical methods are used to detect liquid phase components (both in-process or finished products), measure product purity, or the presence of adulteration or contamination. Aria offers a one-step inexpensive test to detect adulteration or contamination, thus eliminating need for a
presence of adulteration or contamination. Aria offers a one-step inexpensive test to detect adulteration or contamination, thus eliminating need for a combination of measurements from slow and expensive instruments.
combination of measurements from slow and expensive instruments.  The Aria technology has been tested in Quality Assurance applications in a number of industries. Examples of Quality Control applications in Food & Beverages,
The Aria technology has been tested in Quality Assurance applications in a number of industries. Examples of Quality Control applications in Food & Beverages, Inks, Healthcare, and Paints and Coatings, follow:
Inks, Healthcare, and Paints and Coatings, follow:
 presence of adulteration or contamination. Aria offers a one-step inexpensive test to detect adulteration or contamination, thus eliminating need for a
presence of adulteration or contamination. Aria offers a one-step inexpensive test to detect adulteration or contamination, thus eliminating need for a combination of measurements from slow and expensive instruments.
combination of measurements from slow and expensive instruments.  The Aria technology has been tested in Quality Assurance applications in a number of industries. Examples of Quality Control applications in Food & Beverages,
The Aria technology has been tested in Quality Assurance applications in a number of industries. Examples of Quality Control applications in Food & Beverages, Inks, Healthcare, and Paints and Coatings, follow:
Inks, Healthcare, and Paints and Coatings, follow:
Because of their extremely high sensitivity to Brand Reputation issues - Quality Control & Quality Assurance - Food & Beverage producers can see an enormous benefit from using Aria technology. QC and QA are difficult areas for Food & Beverage producers due to unique limitations with respect to controls on raw materials or in-
from using Aria technology. QC and QA are difficult areas for Food & Beverage producers due to unique limitations with respect to controls on raw materials or in- process operations: a large percentage of the raw materials or finished products are imported, some from third world countries.
process operations: a large percentage of the raw materials or finished products are imported, some from third world countries.
 from using Aria technology. QC and QA are difficult areas for Food & Beverage producers due to unique limitations with respect to controls on raw materials or in-
from using Aria technology. QC and QA are difficult areas for Food & Beverage producers due to unique limitations with respect to controls on raw materials or in- process operations: a large percentage of the raw materials or finished products are imported, some from third world countries.
process operations: a large percentage of the raw materials or finished products are imported, some from third world countries.
Quality Control of IV Solutions in Hospital Pharmacies
Aria’s single-measurement technologies can easily distinguish off standard formulations. The test time is equal to the time it takes for
a 2 microliter drop of liquid to dry. The results of such a comparison
are represented in digital format.
The figures at the right show, for example, clear differences
standard formulations. The test time is equal to the time it takes for
a 2 microliter drop of liquid to dry. The results of such a comparison
are represented in digital format.
The figures at the right show, for example, clear differences between inks formulated in volatile vs. aqueous solutions
between inks formulated in volatile vs. aqueous solutions
 standard formulations. The test time is equal to the time it takes for
a 2 microliter drop of liquid to dry. The results of such a comparison
are represented in digital format.
The figures at the right show, for example, clear differences
standard formulations. The test time is equal to the time it takes for
a 2 microliter drop of liquid to dry. The results of such a comparison
are represented in digital format.
The figures at the right show, for example, clear differences between inks formulated in volatile vs. aqueous solutions
between inks formulated in volatile vs. aqueous solutions
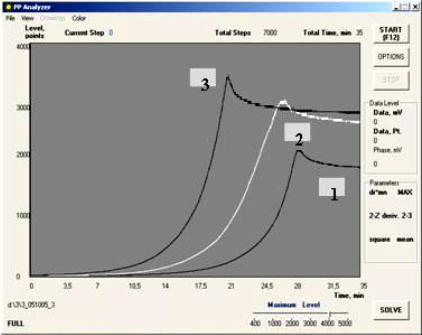
1 – Cranberry Juice;
2 – “False” Orange Juice;
3 – Natural Orange Juice.
Food and Beverage

Food & Beverage products Aria has successfully tested include:
•
Drinking water and ice
•
Milk and dairy products
•
Alcoholic Beverages (e.g. wine, beer)
•
Fruit Juices
•
Condiments
•
Flavors and fragrances
Dairy Products
Juice Authenticity
To Test the Aria Technology, Juice 2 was formulated to have the same physical
formulated to have the same physical appearance and RATIO as natural Orange Juice
appearance and RATIO as natural Orange Juice
 formulated to have the same physical
formulated to have the same physical appearance and RATIO as natural Orange Juice
appearance and RATIO as natural Orange Juice
Inks are a broad set of colloidal systems of fine pigment particles dispersed in an aqueous or organic solvent. In addition to the
dispersed in an aqueous or organic solvent. In addition to the pigment, modern ink formulations contain various amounts of
pigment, modern ink formulations contain various amounts of many other ingredients collectively known as “vehicles”. These
many other ingredients collectively known as “vehicles”. These additional ingredients include pH modifiers, humectants to retard
additional ingredients include pH modifiers, humectants to retard premature drying, polymeric resins to impart binding and allied
premature drying, polymeric resins to impart binding and allied properties, antifoaming agents, wetting agents (control surface
properties, antifoaming agents, wetting agents (control surface properties), biocides to inhibit the fungal and bacterial growth that
lead to fouling, and thickeners or rheology modifiers to control ink
application).
Measurements of many chemical-physical parameters of the ink
properties), biocides to inhibit the fungal and bacterial growth that
lead to fouling, and thickeners or rheology modifiers to control ink
application).
Measurements of many chemical-physical parameters of the ink are required to control ink content and quality.
are required to control ink content and quality.
 dispersed in an aqueous or organic solvent. In addition to the
dispersed in an aqueous or organic solvent. In addition to the pigment, modern ink formulations contain various amounts of
pigment, modern ink formulations contain various amounts of many other ingredients collectively known as “vehicles”. These
many other ingredients collectively known as “vehicles”. These additional ingredients include pH modifiers, humectants to retard
additional ingredients include pH modifiers, humectants to retard premature drying, polymeric resins to impart binding and allied
premature drying, polymeric resins to impart binding and allied properties, antifoaming agents, wetting agents (control surface
properties, antifoaming agents, wetting agents (control surface properties), biocides to inhibit the fungal and bacterial growth that
lead to fouling, and thickeners or rheology modifiers to control ink
application).
Measurements of many chemical-physical parameters of the ink
properties), biocides to inhibit the fungal and bacterial growth that
lead to fouling, and thickeners or rheology modifiers to control ink
application).
Measurements of many chemical-physical parameters of the ink are required to control ink content and quality.
are required to control ink content and quality.
Healthcare
The Agency for Healthcare Research and Quality (U.S. Department of Health and Human Services) has reported that “Medical errors are responsible for injury in as many as 1 out of every 25 hospital patients; an
reported that “Medical errors are responsible for injury in as many as 1 out of every 25 hospital patients; an estimated 48,000-98,000 patients die from medical errors each year. Errors in healthcare have been estimated
estimated 48,000-98,000 patients die from medical errors each year. Errors in healthcare have been estimated to cost more than $5 million per year in a large teaching hospital, and preventable health-care related cost the
economy $17-$29 billion per year.”
These errors may result in a patient receiving the wrong medication, a test result being misread, or even a
to cost more than $5 million per year in a large teaching hospital, and preventable health-care related cost the
economy $17-$29 billion per year.”
These errors may result in a patient receiving the wrong medication, a test result being misread, or even a misdiagnosis. A study by the Agency for Healthcare Research and Quality found that preventable adverse drug
events caused 1 out of 5 injuries or death per year in the hospitals studied. A significant number of the
misdiagnosis. A study by the Agency for Healthcare Research and Quality found that preventable adverse drug
events caused 1 out of 5 injuries or death per year in the hospitals studied. A significant number of the adverse drug events arise is the hospital pharmacy IV room. These errors are among some of the most difficult
to detect and are extremely dangerous to the patient.
adverse drug events arise is the hospital pharmacy IV room. These errors are among some of the most difficult
to detect and are extremely dangerous to the patient.
 reported that “Medical errors are responsible for injury in as many as 1 out of every 25 hospital patients; an
reported that “Medical errors are responsible for injury in as many as 1 out of every 25 hospital patients; an estimated 48,000-98,000 patients die from medical errors each year. Errors in healthcare have been estimated
estimated 48,000-98,000 patients die from medical errors each year. Errors in healthcare have been estimated to cost more than $5 million per year in a large teaching hospital, and preventable health-care related cost the
economy $17-$29 billion per year.”
These errors may result in a patient receiving the wrong medication, a test result being misread, or even a
to cost more than $5 million per year in a large teaching hospital, and preventable health-care related cost the
economy $17-$29 billion per year.”
These errors may result in a patient receiving the wrong medication, a test result being misread, or even a misdiagnosis. A study by the Agency for Healthcare Research and Quality found that preventable adverse drug
events caused 1 out of 5 injuries or death per year in the hospitals studied. A significant number of the
misdiagnosis. A study by the Agency for Healthcare Research and Quality found that preventable adverse drug
events caused 1 out of 5 injuries or death per year in the hospitals studied. A significant number of the adverse drug events arise is the hospital pharmacy IV room. These errors are among some of the most difficult
to detect and are extremely dangerous to the patient.
adverse drug events arise is the hospital pharmacy IV room. These errors are among some of the most difficult
to detect and are extremely dangerous to the patient.
"IV doses are generally mixtures of clear liquids—once mixed, visual inspection of doses simply cannot uncover errors. Human diligence is simply insufficient. In the course of their daily work, hospital pharmacists inspect hundreds, sometimes thousands, of IV
preparations—the vast majority of which are correctly prepared. Finding the rare errors amongst hundreds of correct doses stresses
human capability.”
Dennis Tribble, Chairman of the American Society of Health-Systems Pharmacists; Chief Technology Officer, ForHealth Technologies, Inc.
diligence is simply insufficient. In the course of their daily work, hospital pharmacists inspect hundreds, sometimes thousands, of IV
preparations—the vast majority of which are correctly prepared. Finding the rare errors amongst hundreds of correct doses stresses
human capability.”
Dennis Tribble, Chairman of the American Society of Health-Systems Pharmacists; Chief Technology Officer, ForHealth Technologies, Inc.
 diligence is simply insufficient. In the course of their daily work, hospital pharmacists inspect hundreds, sometimes thousands, of IV
preparations—the vast majority of which are correctly prepared. Finding the rare errors amongst hundreds of correct doses stresses
human capability.”
Dennis Tribble, Chairman of the American Society of Health-Systems Pharmacists; Chief Technology Officer, ForHealth Technologies, Inc.
diligence is simply insufficient. In the course of their daily work, hospital pharmacists inspect hundreds, sometimes thousands, of IV
preparations—the vast majority of which are correctly prepared. Finding the rare errors amongst hundreds of correct doses stresses
human capability.”
Dennis Tribble, Chairman of the American Society of Health-Systems Pharmacists; Chief Technology Officer, ForHealth Technologies, Inc.

Aria testing can be used both in-process and for final IV formulations.
A simple, inexpensive test which requires
for final IV formulations.
A simple, inexpensive test which requires little technical expertise to perform provides
little technical expertise to perform provides a simple straightforward indication of
a simple straightforward indication of whether or not the IV solution has been
whether or not the IV solution has been prepared correctly and is ready for use.
prepared correctly and is ready for use.
 for final IV formulations.
A simple, inexpensive test which requires
for final IV formulations.
A simple, inexpensive test which requires little technical expertise to perform provides
little technical expertise to perform provides a simple straightforward indication of
a simple straightforward indication of whether or not the IV solution has been
whether or not the IV solution has been prepared correctly and is ready for use.
prepared correctly and is ready for use.
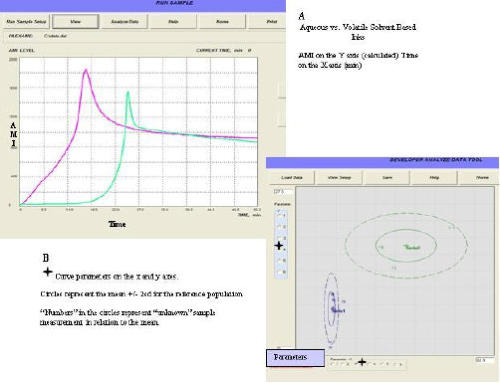
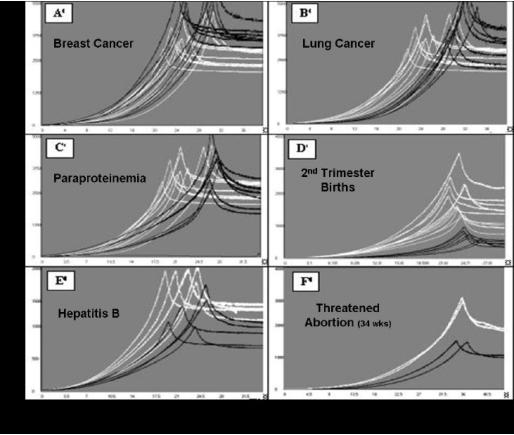
The Aria technology uses a new trend in medical diagnostics - assessment of the morphology of human biological liquids in normal and
disease states. This method provides highly informative rapid results by means of an easy to use, low-cost device.
To evaluate the potential of using this method in medical diagnostics, preliminary studies were performed in Russia with blood plasma
and serum from women in different physiological and pathological states, and with blood serum from patients with paraproteinemia. Additional studies were performed on serum from patients with viral hepatitis, patients who had suffered burns, and women with
Additional studies were performed on serum from patients with viral hepatitis, patients who had suffered burns, and women with breast cancer.
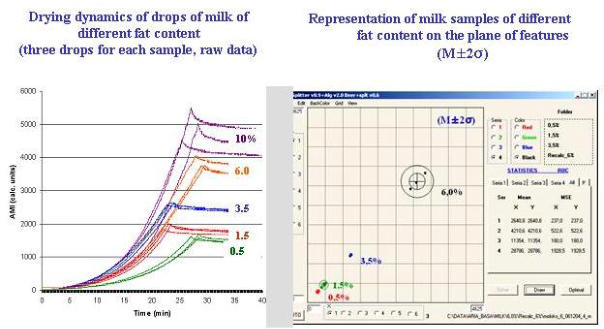
breast cancer.  The figures below show data for healthy patients (data in White) vs patients suffering from various illnesses (data in Black). The clusters
of white vs black data points provide a clear differential between healthy and unhealthy patients.
Aria is seeking partners to help develop these applications more fully.
The figures below show data for healthy patients (data in White) vs patients suffering from various illnesses (data in Black). The clusters
of white vs black data points provide a clear differential between healthy and unhealthy patients.
Aria is seeking partners to help develop these applications more fully.
 Additional studies were performed on serum from patients with viral hepatitis, patients who had suffered burns, and women with
Additional studies were performed on serum from patients with viral hepatitis, patients who had suffered burns, and women with breast cancer.
breast cancer.  The figures below show data for healthy patients (data in White) vs patients suffering from various illnesses (data in Black). The clusters
of white vs black data points provide a clear differential between healthy and unhealthy patients.
Aria is seeking partners to help develop these applications more fully.
The figures below show data for healthy patients (data in White) vs patients suffering from various illnesses (data in Black). The clusters
of white vs black data points provide a clear differential between healthy and unhealthy patients.
Aria is seeking partners to help develop these applications more fully.
Medical Screening

Industrial Applications
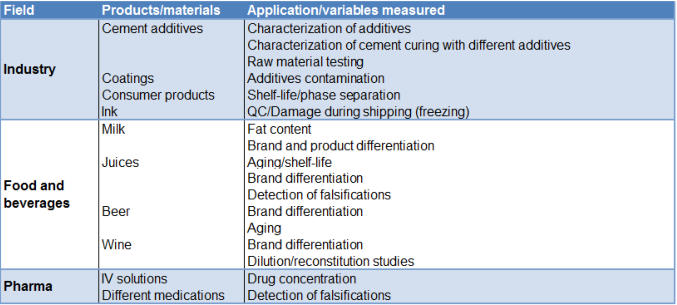
Aria’s platform technology offers numerous practical applications in a variety of industries. Through broad studies the Aria team has shown its potential for many applications. In the table below are just few examples of Aria studies.
Aria team has shown its potential for many applications. In the table below are just few examples of Aria studies.
 Aria team has shown its potential for many applications. In the table below are just few examples of Aria studies.
Aria team has shown its potential for many applications. In the table below are just few examples of Aria studies.

In a similar way to its impact on Quality Assurance, the Aria technology is ideally suited to evaluating Product Performance at or near the
point of sale. The technology can easily measure:
•
Shelf Life
•
Dry Time
•
Cure Time
•
Accurate Mixing
Below are several examples of this type of application:

Polymer Solutions
Paints and Coatings
Aria Technology can easily evaluate samples of different paint batches for variance from standard, both at the factory and at thepoint of sale.
variance from standard, both at the factory and at thepoint of sale.
 variance from standard, both at the factory and at thepoint of sale.
variance from standard, both at the factory and at thepoint of sale.
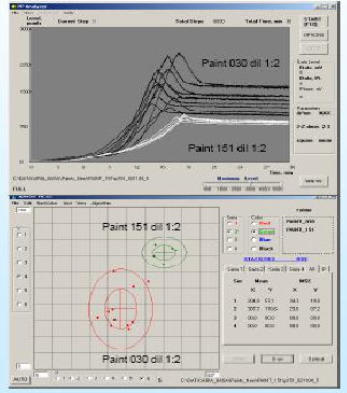
The image at the right shows the results of comparing two different paints against each other. The curves for Paint 151 (in white) show a very stable
against each other. The curves for Paint 151 (in white) show a very stable composition, while the curves for Paint 030 (in black) show changes in the
composition, while the curves for Paint 030 (in black) show changes in the formulation as a function of shelf life.hese results are further emphasized in
formulation as a function of shelf life.hese results are further emphasized in the lower plot, which clearly shows the tight distribution of Paint 151
the lower plot, which clearly shows the tight distribution of Paint 151 compared with the much wider distrbution in data for Paint 030 as a result of
compared with the much wider distrbution in data for Paint 030 as a result of aging.
aging.
 against each other. The curves for Paint 151 (in white) show a very stable
against each other. The curves for Paint 151 (in white) show a very stable composition, while the curves for Paint 030 (in black) show changes in the
composition, while the curves for Paint 030 (in black) show changes in the formulation as a function of shelf life.hese results are further emphasized in
formulation as a function of shelf life.hese results are further emphasized in the lower plot, which clearly shows the tight distribution of Paint 151
the lower plot, which clearly shows the tight distribution of Paint 151 compared with the much wider distrbution in data for Paint 030 as a result of
compared with the much wider distrbution in data for Paint 030 as a result of aging.
aging.

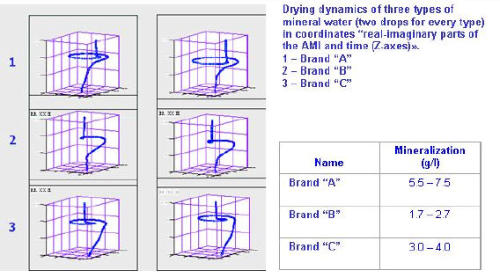
Aria Technology can detect variance from standard in drinking water,
both “from the tap, and bottled waters. The images at the right show
dramatic differences in results from testing samples of mineral water.
water.
 water.
water.
Different levels of mineralization, for example, lead to noticeably different curve shapes for the various samles, allowing for easy
different curve shapes for the various samles, allowing for easy differentiation.
differentiation.
 different curve shapes for the various samles, allowing for easy
different curve shapes for the various samles, allowing for easy differentiation.
differentiation.
Water
Contaminated & Counterfeit Products
In the Chemicals, Food, and Pharmaceuticals areas, as well as in feedstocks for a number of other industries, contamination and counterfeiting are major sources of financial loss.
Contamination can occur at any stage of the supply chain, from raw materials sourcing through manufacturing, through shipping and
counterfeiting are major sources of financial loss.
Contamination can occur at any stage of the supply chain, from raw materials sourcing through manufacturing, through shipping and retail. Of major concern is the rise of global raw material acquisition: companies dramatically increasing joint venture and strategic
retail. Of major concern is the rise of global raw material acquisition: companies dramatically increasing joint venture and strategic partnerships outside the US, and US companies building manufacturing facilities outside the US.
partnerships outside the US, and US companies building manufacturing facilities outside the US.
 counterfeiting are major sources of financial loss.
Contamination can occur at any stage of the supply chain, from raw materials sourcing through manufacturing, through shipping and
counterfeiting are major sources of financial loss.
Contamination can occur at any stage of the supply chain, from raw materials sourcing through manufacturing, through shipping and retail. Of major concern is the rise of global raw material acquisition: companies dramatically increasing joint venture and strategic
retail. Of major concern is the rise of global raw material acquisition: companies dramatically increasing joint venture and strategic partnerships outside the US, and US companies building manufacturing facilities outside the US.
partnerships outside the US, and US companies building manufacturing facilities outside the US.
Fraud & Tampering
Example: Counterfeit Drugs
The current world pharmaceutical market is $541 billion dollars. According to the World Health Organization (WHO), counterfeit drugs make up approximately 7% or $23 billion of the drugs sold into the US market.
The cost to the US healthcare system is huge due to complications, recalls, patient harm, law suits, increased testing, the need for
make up approximately 7% or $23 billion of the drugs sold into the US market.
The cost to the US healthcare system is huge due to complications, recalls, patient harm, law suits, increased testing, the need for otherwise unnecessary patient care, and the cost to “fix the problem.”
To date, only one other method has been identified to test for counterfeit drugs: near IR scanning. Scientists conducting such testing in the
US state that “…each device costs approximately $50,000 and is about the size of a suitcase, making widespread use unfeasible.” Aria’s
otherwise unnecessary patient care, and the cost to “fix the problem.”
To date, only one other method has been identified to test for counterfeit drugs: near IR scanning. Scientists conducting such testing in the
US state that “…each device costs approximately $50,000 and is about the size of a suitcase, making widespread use unfeasible.” Aria’s method offers quick, reliable solution at a fraction of this cost
method offers quick, reliable solution at a fraction of this cost
 make up approximately 7% or $23 billion of the drugs sold into the US market.
The cost to the US healthcare system is huge due to complications, recalls, patient harm, law suits, increased testing, the need for
make up approximately 7% or $23 billion of the drugs sold into the US market.
The cost to the US healthcare system is huge due to complications, recalls, patient harm, law suits, increased testing, the need for otherwise unnecessary patient care, and the cost to “fix the problem.”
To date, only one other method has been identified to test for counterfeit drugs: near IR scanning. Scientists conducting such testing in the
US state that “…each device costs approximately $50,000 and is about the size of a suitcase, making widespread use unfeasible.” Aria’s
otherwise unnecessary patient care, and the cost to “fix the problem.”
To date, only one other method has been identified to test for counterfeit drugs: near IR scanning. Scientists conducting such testing in the
US state that “…each device costs approximately $50,000 and is about the size of a suitcase, making widespread use unfeasible.” Aria’s method offers quick, reliable solution at a fraction of this cost
method offers quick, reliable solution at a fraction of this cost
This figure shows the identification of the counterfeit drug in less than 15 minutes.
This drug is an injectable polypeptide manufactured in Austria for the treatment of Alzheimer’s disease and ischemic stroke. It has been approved for use in 44 countries
and is in pre-clinical testing in the US.
This counterfeit has already entered the market.
Alzheimer’s disease and ischemic stroke. It has been approved for use in 44 countries
and is in pre-clinical testing in the US.
This counterfeit has already entered the market.
 Alzheimer’s disease and ischemic stroke. It has been approved for use in 44 countries
and is in pre-clinical testing in the US.
This counterfeit has already entered the market.
Alzheimer’s disease and ischemic stroke. It has been approved for use in 44 countries
and is in pre-clinical testing in the US.
This counterfeit has already entered the market.
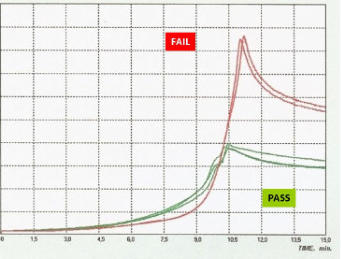
The figure shows the raw data for a “good” lot of product (Green) with a counterfeit lot (Red).
The Aria technology clearly distinguishes the difference.
lot (Red).
The Aria technology clearly distinguishes the difference.
 lot (Red).
The Aria technology clearly distinguishes the difference.
lot (Red).
The Aria technology clearly distinguishes the difference.
Counterfeiting, on the other hand, is a purposeful attempt to defraud the buyer. Four basic categories of counterfeits are:
•
Completely fraudulent counterfeit products with no active ingredients or benefit.
•
Counterfeit products which contain poor-quality ingredients or a diluted active ingredient.
•
Expired products that are re-packaged and resold.
•
Products that mimic the proprietary formula using unknown raw materials/ingredients and manufacturing processes.
Aria’s solution allows for rapid detection of deviation from standard formulations with contaminated and counterfeit products just as it does its QC applications in manufacturing:
does its QC applications in manufacturing:
 does its QC applications in manufacturing:
does its QC applications in manufacturing:
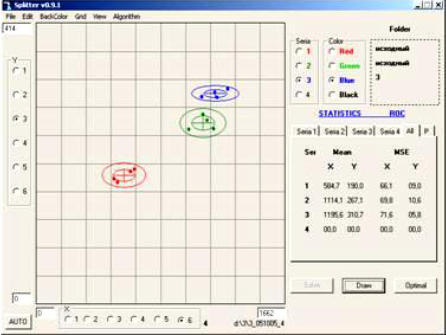
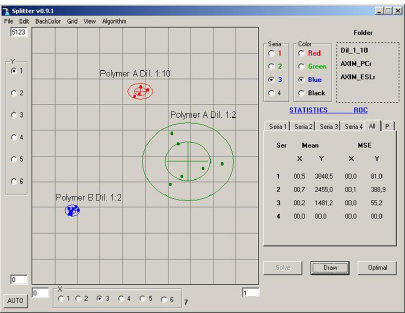
Aria provides a way to detect deviations from standard additives (or final products) in a single measurement. The figure to the right show an example of
products) in a single measurement. The figure to the right show an example of various dilution levels for an aqueous solution of a proprietary polymer. The red
and green clusters represent two dilutions of the same polymer (“A”). The blue
various dilution levels for an aqueous solution of a proprietary polymer. The red
and green clusters represent two dilutions of the same polymer (“A”). The blue cluster represents a solution of a second polymer (“B”) at the same 1:2 dilution as
cluster represents a solution of a second polymer (“B”) at the same 1:2 dilution as polymer “A” in green. The clusters are clearly distinguishable from each other.
polymer “A” in green. The clusters are clearly distinguishable from each other.
 products) in a single measurement. The figure to the right show an example of
products) in a single measurement. The figure to the right show an example of various dilution levels for an aqueous solution of a proprietary polymer. The red
and green clusters represent two dilutions of the same polymer (“A”). The blue
various dilution levels for an aqueous solution of a proprietary polymer. The red
and green clusters represent two dilutions of the same polymer (“A”). The blue cluster represents a solution of a second polymer (“B”) at the same 1:2 dilution as
cluster represents a solution of a second polymer (“B”) at the same 1:2 dilution as polymer “A” in green. The clusters are clearly distinguishable from each other.
polymer “A” in green. The clusters are clearly distinguishable from each other.
A major ingredient in many products starts as a solution of various polymers. Polymer solutions are also commnon additives for non-polymer products. These
Polymer solutions are also commnon additives for non-polymer products. These constituents are used in everything from paints, coatings, priniting, and packaging
products to automotive, electronics, medical, and pharmaceutical applications.
constituents are used in everything from paints, coatings, priniting, and packaging
products to automotive, electronics, medical, and pharmaceutical applications. Screening of incoming raw materials can catch problems with feedstock, but
Screening of incoming raw materials can catch problems with feedstock, but testing usuallly requires a combination of measurements from slow and
testing usuallly requires a combination of measurements from slow and expensive instruments.
expensive instruments.
 Polymer solutions are also commnon additives for non-polymer products. These
Polymer solutions are also commnon additives for non-polymer products. These constituents are used in everything from paints, coatings, priniting, and packaging
products to automotive, electronics, medical, and pharmaceutical applications.
constituents are used in everything from paints, coatings, priniting, and packaging
products to automotive, electronics, medical, and pharmaceutical applications. Screening of incoming raw materials can catch problems with feedstock, but
Screening of incoming raw materials can catch problems with feedstock, but testing usuallly requires a combination of measurements from slow and
testing usuallly requires a combination of measurements from slow and expensive instruments.
expensive instruments.








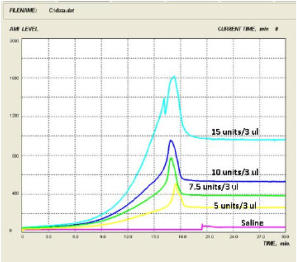
Varying Concentrations of Heparin

Countless Other
Applications ...